Product information
Natural fire-retardant mineral
UltraCarb is a cost-effective mixture of minerals, which is also known as HMH. It is a proven fire-retardant filler, used in a wide range of polymers and rubbers such as polyethylene (PE), polypropylene (PP), polyvinyl chloride (PVC), ethylene-propylene diene monomer (EPDM) and nitrile rubber (NBR).
The combination gives a unique product in which specific properties can be optimised by selected mining and processing to fit a variety of applications.
Unique fire retardant properties
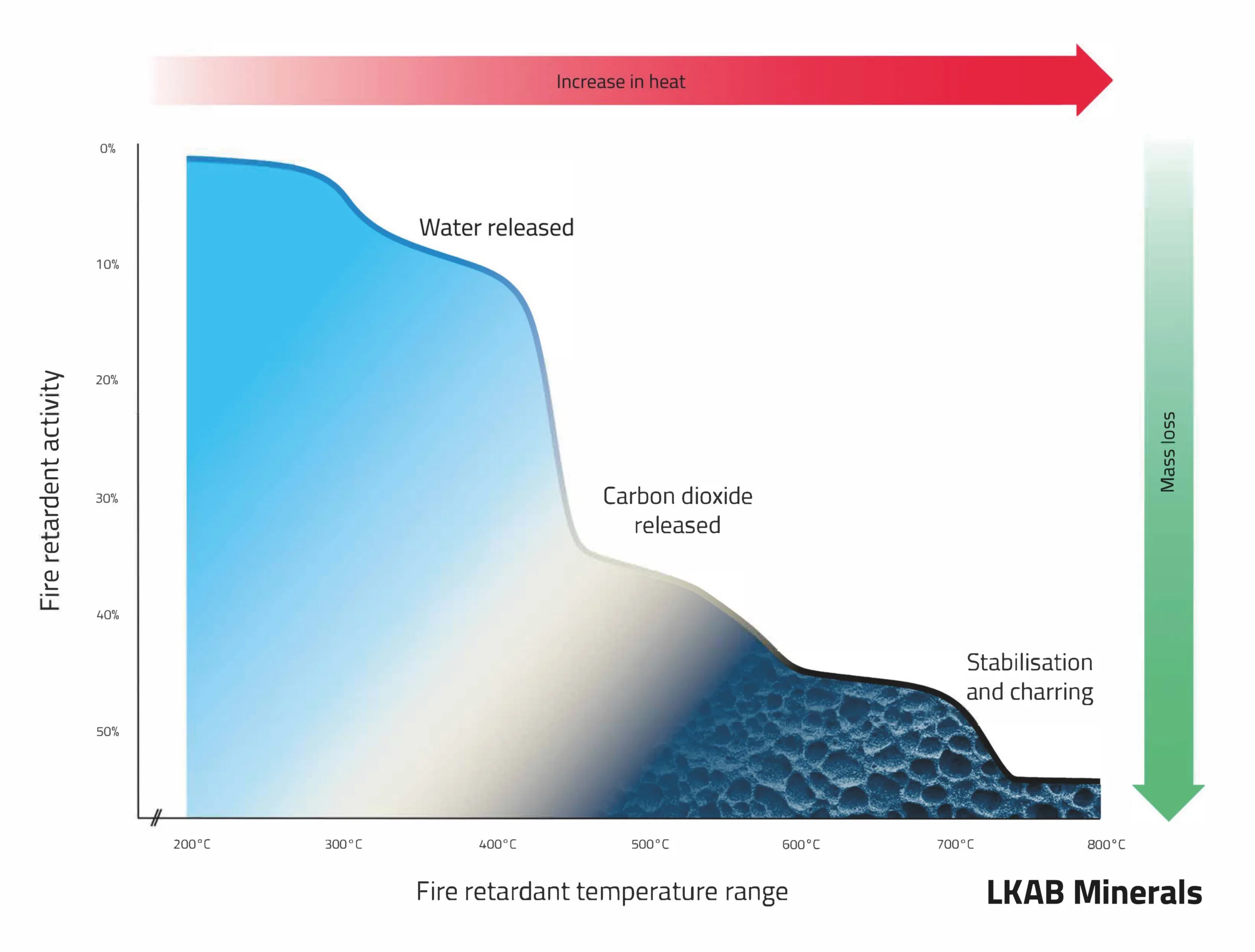
UltraCarb has a unique 3-stage fire-retardant mechanism. Whereas the classical endothermic flame retardant fillers act by releasing water in a single action process, our product works as follows:
- Water is released at temperatures of ~220°C (thermal decomposition)
- CO 2 release at temperatures of ~330°C
- At temperatures of ~560°C, particles accumulate at the surface of the burning polymer to form a stable cementitious char. This char inhibits the combustion process and thus the propagation of the fire through burning droplets.
The platy, high surface area particles of our halogen-free mineral provide excellent properties when you use these as a functional filler in polymers. The products’ high heat absorption protects polymers from rapid thermal degradation and the formation of combustible products.
UltraCarb standard grades
| Primary Hydromagnesite | Primary Huntite |
|---|---|
| LH3 | 1250 |
| LH15 | 1251 (stearate surface treated) |
| LH15C (stearate surface treated) | 1253 (proprietary surface treated) |
| LH15X (proprietary surface treated) | 1290 |
| 1291 (proprietary surface treated) |
UltraCarb packaging
Our UltraCarb product range is available in paper sacks, big bags or in bulk.
Stearate surface treatment improves wetting and reduces the agglomeration in the compound; proprietary surface treatments are beneficial as an external lubricant, act as a processing aid for extrusion and improve the LOI in EVA.
Mineral information
The typical properties of UltraCarb are:
Physical properties of UltraCarb
| Specific gravity [g/cm3] | 2.4 |
| Refractive index | 1.56 |
| Hardness (Mohs) | 2.5 |
| Surface area [m2/g] | 11 – 17 |
| Loose bulk density [kg/l] | 0.4 |
Chemical properties of UltraCarb
| MgO [%] | 36 – 39 |
| CaO [%] | 6 – 9 |
| SiO2 [%] | 0.2 – 1 |
| Loss on ignition at 1,000°C | 51 – 54 |
UltraCarb origin
The naturally occurring mixtures of Hydromagnesite and Huntite mineral deposits are formed by near surface geochemical processes. The formation of our own Turkish deposits is the result of three specific conditions:
- a depositional area of magnesium rich ultramafic basement rocks;
- a lake environment which evolved into a basin;
- tectonic activity: This allowed magnesium rich ground water to reach the surface.
The precipitation of HMH is related to the mixing of ground water with fresh water from the lake environment.
HMH toxicology
UltraCarb is naturally halogen-free, therefore does not emit toxic smoke when burnt.

